“Game-changing" Thermometer by TriMedika to prevent cross-infection
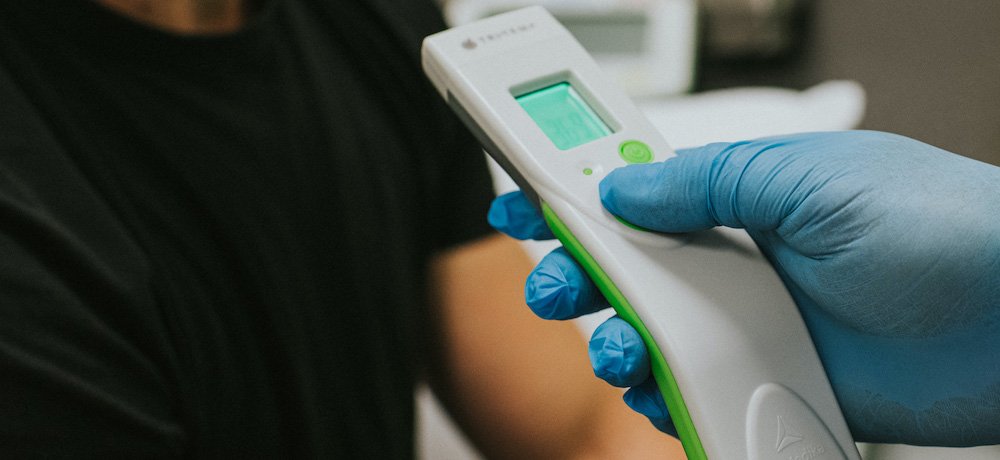
TriMedika, a medical technology manufacturer providing clinical devices for hospitals worldwide, showcased its “game-changing” thermometer at the ABHI UK Pavilion at MEDICA 2021. the purpose of the thermometer is to prevent cross-infection in hospitals.
TriMedika’s vision is to improve patient care globally with technology that protects patients, which is inspired by first-hand experience of the challenges of healthcare and applying fresh thinking to solve simple yet critical everyday issues,
As such, the company’s top product TRITEMP, a medically graded professional non-contact thermometer is a precision engineered tool designed to positively impact patient care and control infection across healthcare globally.
In a time when there was a clear need for the product before the pandemic, the COVID-19 crisis has further highlighted the importance of infection risks associated with contact.
“There have been very few developments within the thermometry industry for many years. So, we set about designing a non-contact infrared thermometer, accurate and robust enough to stand up to use in busy hospital wards,” COO Julie Brien said.
“The benefits in terms of infection control and improved patient comfort are significant,” he noted.
Over 1000 hospitals globally, including specialist units such as neonatal wards and ICU’s, have switched to TRITEMP. Hospitals can gain many benefits across staffing, workflows and costs, by introducing the device.
TriMedika is supplied to hospitals in 21 countries through its distribution partners, in parallel with planning to enter the U.S. market in 2022.
The new device is shortlisted for Disruptor of the Year at the Great British Entrepreneur Awards 2021. CEO Dr Roisin Molloy is also in the running for the prestigious EY Entrepreneur of the Year.
Paul Benton, managing director, International at ABHI said: “We’re excited that TriMedika have chosen to showcase their innovative solution on the ABHI Pavilion, where they’re joined by some of the most exciting and forward-thinking UK companies across the medical world. We look forward to welcoming them.”
